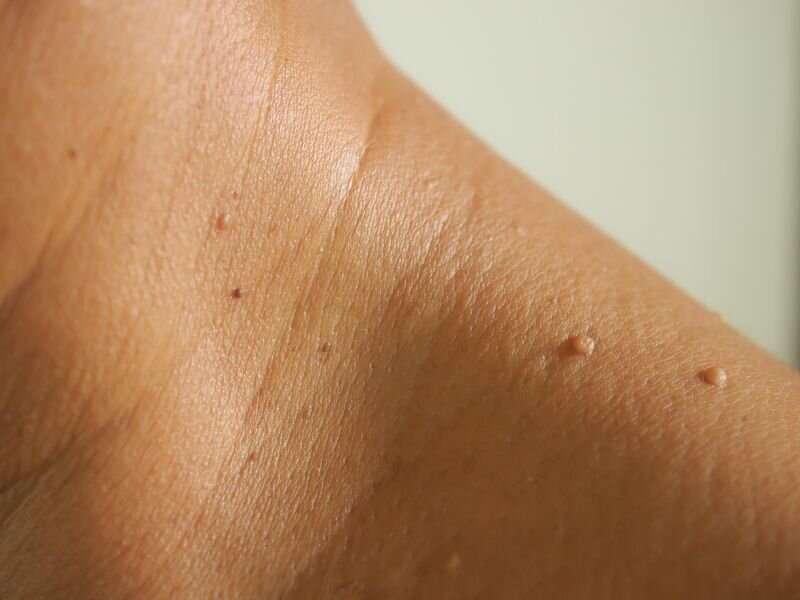
The U.S. Meals and Drug Administration on Tuesday issued warning letters to 3 firms, together with Amazon, for promoting unapproved merchandise for eradicating moles and pores and skin tags.
No over-the-counter medicines have FDA approval for that goal, and the Meals, Drug, and Beauty Act prohibits interstate sale of unapproved medicine and cosmetics.
“It’s the FDA’s obligation to guard public well being from dangerous merchandise not permitted for the U.S. market,” mentioned Donald Ashley, director of the compliance workplace within the FDA’s Heart for Drug Analysis and Analysis. “This consists of the place on-line retailers like Amazon are concerned within the interstate sale of unapproved drug merchandise. We are going to proceed to work diligently to make sure that on-line retailers don’t promote merchandise that violate federal regulation.”
In addition to Amazon, the FDA issued warning letters to Ariella Naturals and Justified Laboratories.
The company emphasised that it has not examined the mole and pores and skin tag removing merchandise offered by these firms for security, effectiveness or high quality.
The warning letters put the businesses on discover that failure to deal with the violations cited by the FDA could end in authorized motion, together with seizure. As soon as they obtain the warning letter, the businesses have 15 days to tell the company of any corrective measures they’ve taken.
The company warned earlier this summer time that utilizing unregulated merchandise for do-it-yourself removing of moles, pores and skin tags and one other sort of progress generally known as seborrheic keratoses can masks pores and skin most cancers and end in wounds and scarring.
“There are a number of causes that sufferers ought to keep away from making an attempt to deal with moles at house. And that’s actually essentially the most regarding… that most cancers sufferers typically mistake pores and skin most cancers for benign moles,” Dr. Chad Prather, a board-certified dermatologist in Baton Rouge, La, mentioned on the time. “We generally see sufferers who’ve a pores and skin most cancers. It has been recognized and their preliminary course of was to attempt to deal with that at house with both bodily means or generally these over-the-counter merchandise.”
That may cloud the analysis of very severe pores and skin cancers, resembling melanoma, Prather mentioned.
“We decide how unhealthy a melanoma is by how deep it goes. And we actually want that preliminary biopsy to know the true depth in order that we will select essentially the most acceptable remedy methodology, whether or not it is surgical procedure or checking lymph nodes or adopted by immunotherapy,” Prather defined.
The FDA mentioned Tuesday that it’ll proceed to make use of all of the instruments out there to guard public well being and take away fraudulent or unproven drug merchandise from the American market. The warning letters had been simply step one.
Customers and medical professionals are requested to report any hostile occasions associated to the merchandise to the MedWatch Hostile Occasion Reporting.
BeSafeRx is a U.S. Meals and Drug Administration website for on-line pharmacy info.
Copyright © 2022 HealthDay. All rights reserved.
Quotation:
FDA warns Amazon, different distributors about sale of pores and skin tag removing merchandise (2022, August 10)
retrieved 13 August 2022
from https://medicalxpress.com/information/2022-08-fda-amazon-vendors-sale-skin.html
This doc is topic to copyright. Other than any honest dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.




