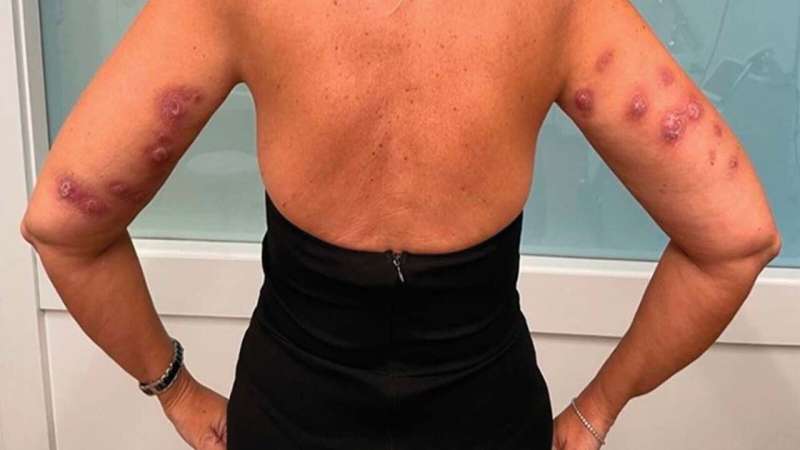
Individuals are being maimed by unauthorized fat-dissolving injections meant to tighten up double chins and dissipate flab alongside the arms, thighs and abdomen, the U.S. Meals and Drug Administration warns.
The photographs are supposed to interrupt down fats cells and cut back fats deposits within the areas across the injection websites.
However adversarial reactions from the unapproved injections are inflicting scarring, pores and skin deformities, cysts, painful knots, and critical infections, the FDA stated in an company information launch.
The unapproved injections are being marketed underneath model names like Aqualyx, Lipodissolve, Lipo Lab and Kabelline, the FDA stated.
Frequent components within the injections embody phosphatidylcholine (PPC) and sodium deoxycholate (DC). These components have been used alone or collectively and are generally known as “PCDC injections.”
The FDA has acquired studies of shoppers harmed by injections they acquired at clinics or med spas from attendants who may not have been correctly licensed to present the photographs.
The company additionally has heard from some shoppers who purchased the unapproved photographs on-line and injected the medication themselves.
The FDA has permitted just one injectable drug for dissolving fats, a prescription treatment referred to as Kybella, the company stated.
The drug, which is deozycholic acid, is FDA-approved to deal with double chins in adults. Deozycholic acid is a bile acid naturally produced by intestinal micro organism to assist break down fat throughout digestion.
The FDA warns that improper or unsafe injection practices can improve the chance of scarring, pores and skin infections and critical problems.
Protected and efficient use of a fat-dissolving agent entails calculating the proper quantity and placement of the injections, inserting the needles correctly, and administering the photographs in a protected and sterile method, the FDA stated.
Kybella’s label notes that the photographs ought to be administered solely by a well being care skilled, the company famous.
Folks should not purchase fat-dissolving merchandise from web sites, a lot much less try and inject them, the FDA added.
As an alternative, they need to seek the advice of their physician about FDA-approved remedies like Kybella.
People who’ve acquired these injections and are experiencing unwanted effects ought to search medical care. In addition they ought to report their case to the FDA’s MedWatch program, which helps the company monitor treatment questions of safety.
Extra info:
The American Academy of Dermatology has extra about noninvasive fats removing.
Copyright © 2023 HealthDay. All rights reserved.
Quotation:
Unapproved ‘fat-dissolving’ injections are leaving sufferers maimed, FDA warns (2023, December 22)
retrieved 23 December 2023
from https://medicalxpress.com/information/2023-12-unapproved-fat-dissolving-patients-maimed-fda.html
This doc is topic to copyright. Other than any honest dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.




